Non-small cell lung cancer (NSCLC) with epidermal growth factor receptor (EGFR) exon 20 insertion mutations represents a biologically distinct subset of lung adenocarcinoma, accounting for approximately 4% to 12% of EGFR-mutated NSCLC cases. Unlike classical sensitizing mutations such as exon 19 deletions and L858R, exon 20 insertions are often resistant to first- and second-generation EGFR tyrosine kinase inhibitors (TKIs), necessitating dedicated therapeutic strategies.
Molecular Profile of EGFR Exon 20 Insertions
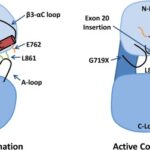
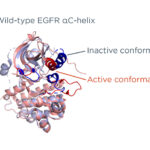
EGFR exon 20 insertion mutations result from in-frame insertions or duplications of 3–21 base pairs, occurring after codon 764 through 774. These changes lead to structural alterations in the EGFR kinase domain that impair binding of traditional EGFR-TKIs while maintaining kinase activity, thus promoting oncogenic signaling through MAPK, PI3K/AKT, and JAK/STAT pathways.
These molecular changes confer intrinsic resistance to earlier EGFR-targeted agents, making this subtype particularly challenging to treat.
Epidemiology and Clinical Characteristics
- Prevalence: 1%–2% of all NSCLC cases; 4%–12% among EGFR-mutated NSCLC
- Histology: Primarily adenocarcinoma
- Demographics:
- More common in non-smokers
- Higher prevalence in Asian populations
- Slight predominance in female patients
Unlike classic EGFR mutations, exon 20 insertions lack a uniform response to conventional EGFR inhibition, underscoring the need for mutation-specific interventions.
Diagnostic Approaches for EGFR Exon 20 Insertions
Tissue-Based Genomic Testing
- Next-generation sequencing (NGS) is the most accurate method to identify the diverse and heterogeneous exon 20 insertion variants
- RT-PCR and Sanger sequencing are limited by detection sensitivity and scope
- Precise mutation identification is essential, as therapeutic efficacy may vary by insertion subtype
Liquid Biopsy (ctDNA Analysis)
- Valuable when tissue is insufficient or inaccessible
- Enables detection of resistance mechanisms and dynamic tumor evolution
Accurate genomic profiling must be integrated into the diagnostic workflow for all advanced NSCLC cases.
Limitations of Conventional EGFR-TKIs in EGFR Exon 20 Insertions
First- and second-generation EGFR-TKIs (gefitinib, erlotinib, afatinib) demonstrate limited to no activity against EGFR exon 20 insertions due to poor drug-receptor affinity and high ATP competition.
Resistance Mechanism
- The P-loop and C-helix structural changes resulting from exon 20 insertions hinder TKI binding
- High ATP affinity of mutant EGFR prevents effective competitive inhibition
This intrinsic resistance necessitated the development of novel agents specifically engineered to overcome the unique conformation of exon 20 mutant EGFR.
Targeted Therapies for EGFR Exon 20 Insertion NSCLC
Amivantamab-vmjw (Rybrevant®)
A bispecific monoclonal antibody targeting both EGFR and MET, approved for patients with locally advanced or metastatic NSCLC harboring EGFR exon 20 insertion mutations post-platinum chemotherapy.
Key Findings from CHRYSALIS Trial
- Objective Response Rate (ORR): 40%
- Median Progression-Free Survival (PFS): 8.3 months
- Median Overall Survival (OS): 22.8 months
Amivantamab is administered intravenously and is associated with:
- Infusion-related reactions (most common adverse event)
- Rash and paronychia
Mobocertinib (Exkivity™)
An oral EGFR TKI designed specifically for exon 20 insertions. FDA-approved for patients with previously treated EGFR exon 20 insertion-mutated NSCLC.
EXCLAIM Study Results
- ORR: 28%
- PFS: 7.3 months
- OS: 24 months
Common toxicities include:
- Diarrhea
- Rash
- QT prolongation
Mobocertinib offers a convenient oral option but may require dose adjustments for toxicity management.
Emerging Therapies and Combination Approaches
Sunvozertinib (DZD9008)
- Next-generation EGFR-TKI with activity against multiple exon 20 variants
- Promising early-phase results with improved selectivity
CLN-081 (TAS6417)
- Oral agent showing preclinical activity in various exon 20 insertion subtypes
- Under evaluation in multiple trials
Combination Strategies
- Amivantamab + lazertinib
- EGFR TKIs + MET inhibitors (e.g., tepotinib)
- Anti-angiogenic agents + chemotherapy
Comparison of Targeted Therapies in EGFR Exon 20 Insertion NSCLC
| Therapy | Type | ORR | PFS (months) | Administration | Approved Setting |
|---|---|---|---|---|---|
| Amivantamab | EGFR/MET Bispecific | ~40% | ~8.3 | IV | Post-chemotherapy |
| Mobocertinib | EGFR-TKI (Oral) | ~28% | ~7.3 | Oral | Post-chemotherapy |
| CLN-081 | EGFR-TKI (Investigational) | ~41% | ~10 (early data) | Oral | Clinical trials only |
| Sunvozertinib | EGFR-TKI (Selective) | ~52% | ~12 (preliminary) | Oral | Clinical trials only |
Treatment Sequencing and Clinical Considerations
First-Line Therapy
- Platinum-doublet chemotherapy remains standard first-line treatment
- Inclusion of bevacizumab or immune checkpoint inhibitors evaluated case-by-case
Second-Line and Beyond
- Amivantamab or mobocertinib for exon 20-mutated disease progression
- Consider clinical trial enrollment for investigational exon 20-targeted agents
Monitoring and Follow-Up
- Imaging every 6–12 weeks based on disease kinetics
- ctDNA testing at progression to detect acquired resistance
- Surveillance for CNS involvement, as exon 20 NSCLC may develop brain metastases
Challenges in Immunotherapy for EGFR Exon 20 Insertion NSCLC
Immune checkpoint inhibitors (ICIs) have limited efficacy in EGFR-mutated NSCLC, including exon 20 insertion variants.
- Low tumor mutational burden (TMB)
- Reduced PD-L1 expression
- Lack of immune cell infiltration (cold tumors)
ICIs may have a role post-TKI failure in combination with chemotherapy, particularly in high PD-L1 expressing tumors.
Prognostic Implications
NSCLC patients with EGFR exon 20 insertions generally exhibit poorer prognosis than those with classical EGFR mutations due to:
- Lack of early targeted options until recent approvals
- Limited efficacy of conventional TKIs
- Higher likelihood of disease progression under chemotherapy
However, with novel agents such as amivantamab and mobocertinib, outcomes are improving, and survival benchmarks are approaching those seen in classical EGFR-mutant disease.
Non-small cell lung cancer with EGFR exon 20 insertion mutations presents unique challenges in molecular targeting due to intrinsic resistance to earlier EGFR inhibitors. The advent of mutation-specific therapies such as amivantamab and mobocertinib has significantly advanced treatment outcomes. With ongoing clinical trials investigating more selective and potent inhibitors, the future holds promise for improving survival and quality of life in this hard-to-treat population. Continued innovation in diagnostic accuracy, personalized treatment, and resistance monitoring will be critical to optimizing care for patients with EGFR exon 20 insertion NSCLC.