Non CNS Manifestations of Acid Sphingomyelinase Deficiency: Acid sphingomyelinase deficiency (ASMD) is a rare lysosomal storage disorder caused by mutations in the SMPD1 gene, resulting in reduced or absent activity of the acid sphingomyelinase (ASM) enzyme. This deficiency leads to the accumulation of sphingomyelin and other lipids in various tissues, particularly within the reticuloendothelial system, affecting multiple organs. While central nervous system (CNS) involvement characterizes the severe infantile neurovisceral form (Niemann-Pick disease type A), this article focuses on non-CNS manifestations, particularly relevant in chronic visceral (type B) and intermediate forms of the disease.
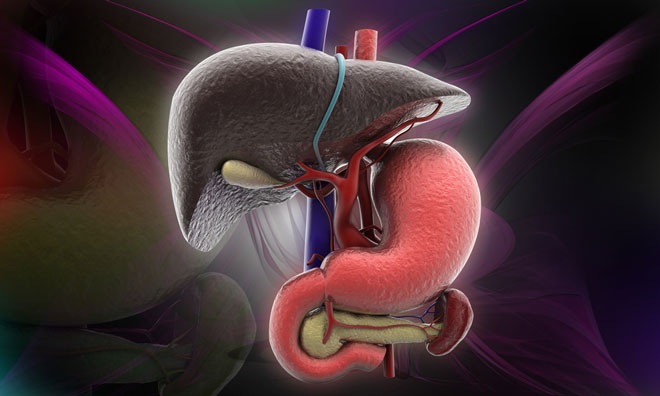
Visceral Manifestations: Hepatosplenomegaly and Liver Dysfunction
Hepatomegaly and Splenomegaly
Enlargement of the liver and spleen is one of the earliest and most consistent findings in ASMD. The pathologic accumulation of sphingomyelin-laden macrophages (foam cells) within these organs leads to:
- Hepatomegaly: Liver enlargement is often massive, with patients experiencing abdominal distension and discomfort. Liver enzymes (ALT, AST) may be mildly elevated.
- Splenomegaly: Splenic involvement is often more pronounced than hepatic, contributing to hypersplenism, which exacerbates cytopenias.
Liver Dysfunction
Progressive lipid accumulation may cause hepatic fibrosis, and in advanced cases, portal hypertension and cirrhosis. However, frank liver failure is uncommon. Liver biopsy often reveals foam cells and fibrosis but not the severe inflammation seen in viral or alcoholic liver disease.
Hematologic Abnormalities: Cytopenias and Coagulation Disorders
Anemia and Thrombocytopenia
Secondary to hypersplenism, many patients develop:
- Anemia: Normocytic, normochromic anemia is common, contributing to fatigue.
- Thrombocytopenia: Low platelet counts are a hallmark, often leading to easy bruising, epistaxis, and prolonged bleeding after minor trauma.
Leukopenia and Bleeding Risk
Though less common, leukopenia may predispose individuals to infections. Coagulopathies may arise from both platelet dysfunction and hepatocellular impairment, increasing the risk of spontaneous bleeding.
Pulmonary Involvement: Interstitial Lung Disease and Restrictive Defects
Pulmonary manifestations are prevalent and often insidious, progressing over time to cause substantial morbidity.
Interstitial Lung Disease (ILD)
- Lipid-laden macrophages infiltrate the alveolar spaces and interstitium, resulting in diffuse pulmonary involvement.
- Radiographic Findings: High-resolution CT scans frequently reveal ground-glass opacities, reticulonodular patterns, and septal thickening.
Respiratory Function Impairment
- Restrictive lung disease with reduced total lung capacity (TLC) and diffusing capacity for carbon monoxide (DLCO) is common.
- Symptoms include exertional dyspnea, dry cough, and progressive hypoxemia, particularly during physical activity.
Cardiovascular Manifestations: Atherogenic Risk and Valvular Involvement
Dyslipidemia
ASMD patients exhibit a highly atherogenic lipid profile:
- Elevated low-density lipoprotein cholesterol (LDL-C)
- Increased triglycerides
- Reduced high-density lipoprotein cholesterol (HDL-C)
This dyslipidemia contributes to early-onset atherosclerosis, raising concerns for coronary artery disease (CAD) and cerebrovascular complications.
Cardiac Infiltration
Although less frequent, myocardial and valvular involvement has been reported. Echocardiographic findings may include:
- Valvular thickening, particularly of the mitral and aortic valves
- Left ventricular hypertrophy due to infiltrative cardiomyopathy
Skeletal Abnormalities: Growth Impairment and Bone Density
Growth Retardation
Children with ASMD often display short stature, which may persist into adulthood. Contributing factors include:
- Chronic illness
- Nutritional compromise from hepatosplenomegaly
- Hormonal dysregulation
Bone Marrow and Density
- Bone marrow infiltration by lipid-laden macrophages may contribute to hematologic abnormalities.
- Osteopenia or osteoporosis can develop, increasing the risk of fractures, especially in older patients.
Gastrointestinal and Abdominal Complications
Gastrointestinal Symptoms
- Abdominal pain and fullness from organomegaly
- Diarrhea or steatorrhea in cases with significant lipid malabsorption
- Rarely, intestinal lipid deposits may cause mucosal dysfunction
Nutritional Impact
Chronic abdominal discomfort and early satiety may reduce caloric intake, exacerbating failure to thrive in pediatric cases and malnutrition in adults.
Dermatologic and Ocular Features
Though not universal, some patients display non-CNS-related skin and eye findings:
- Xanthomas due to lipid accumulation
- Cherry-red macula in some non-neuronopathic cases, though more characteristic of type A
- Corneal opacities may occur due to lipid deposition, though vision remains largely unaffected
Clinical Monitoring and Diagnostic Workup
Routine Assessments
To monitor non-CNS involvement, regular assessments are crucial:
- Liver function tests and abdominal ultrasound
- Complete blood count for cytopenias
- Pulmonary function tests and chest imaging
- Lipid panels for atherogenic risk
- Echocardiography to assess cardiac function
Biomarkers and Genetic Testing
- Plasma lysosphingomyelin levels can serve as a disease biomarker
- Confirmatory diagnosis via SMPD1 gene sequencing and ASM enzyme assay
Management Strategies for Non-CNS Manifestations
Enzyme Replacement Therapy (ERT)
Olipudase alfa, an investigational recombinant human ASM, has shown promise in reducing spleen and liver size, improving pulmonary function, and ameliorating lipid profiles.
Supportive Care
- Blood transfusions for anemia
- Supplemental oxygen for pulmonary impairment
- Statins and dietary control for lipid abnormalities
- Growth hormone therapy (select cases)
Multidisciplinary Approach
Effective care requires coordinated management by:
- Hematologists
- Pulmonologists
- Hepatologists
- Cardiologists
- Metabolic disease specialists
Prognosis and Quality of Life
While ASMD is a lifelong condition, early identification and targeted management of non-CNS manifestations significantly enhance quality of life, reduce complications, and improve functional outcomes. Advancements in enzyme replacement therapy mark a turning point in altering disease progression and optimizing systemic health.