Myelodysplastic syndrome (MDS) is a heterogeneous group of bone marrow disorders characterized by ineffective hematopoiesis and a risk of progression to acute myeloid leukemia (AML). A subset of MDS patients carries mutations in the isocitrate dehydrogenase-1 (IDH1) gene, which alters cellular metabolism and contributes to leukemogenesis. Identifying and targeting the IDH1 mutation has become a critical component of modern MDS management.
Molecular Pathophysiology of IDH1 Mutation in MDS
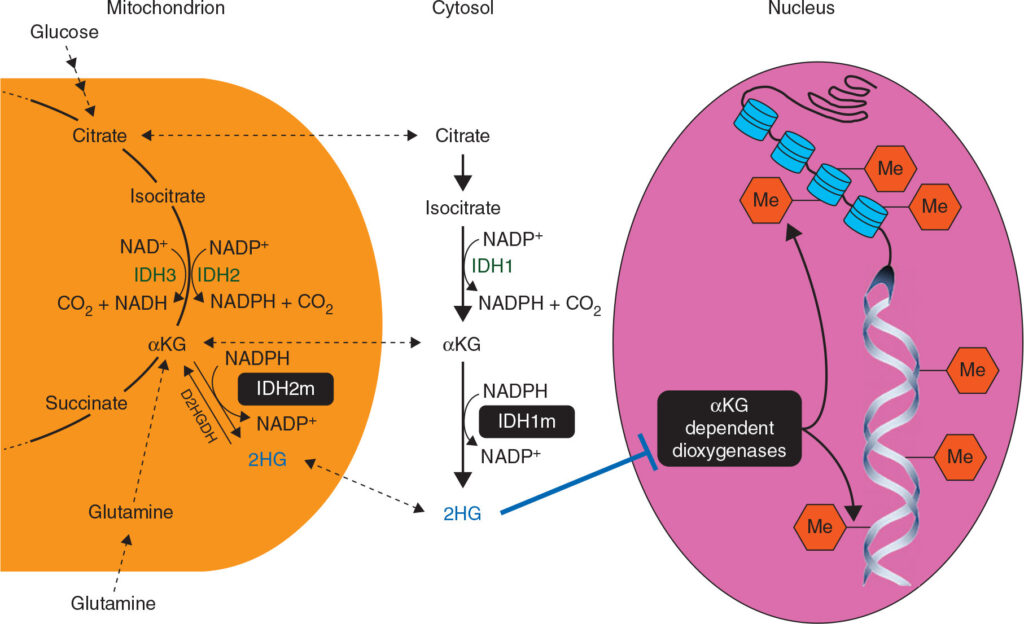
IDH1 is an enzyme involved in the citric acid cycle, converting isocitrate to α-ketoglutarate (α-KG). In mutated IDH1, this function is altered, leading to the production of D-2-hydroxyglutarate (D-2HG), an oncometabolite that disrupts DNA methylation and differentiation.
This dysregulation leads to abnormal blood cell production, increased blast accumulation, and progression to AML.
Causes and Risk Factors
The IDH1 mutation occurs as an acquired genetic event in MDS. While its exact cause is not fully understood, several risk factors contribute to its development:
- Aging – IDH1 mutations are more common in older patients.
- Genotoxic Exposure – Prior chemotherapy, radiation, or benzene exposure increases the risk.
- Genetic Susceptibility – Clonal hematopoiesis of indeterminate potential (CHIP) may predispose individuals to mutations.
- Bone Marrow Stress – Chronic inflammation and oxidative stress may contribute to IDH1 alterations.
Symptoms of Myelodysplastic Syndrome with IDH1 Mutation
The clinical presentation of IDH1-mutated MDS is similar to other forms of MDS but may have distinct features:
- Fatigue and Pallor – Due to anemia from ineffective erythropoiesis.
- Easy Bruising or Bleeding – Resulting from thrombocytopenia.
- Infections – Increased susceptibility due to neutropenia.
- Neurological Symptoms – In some cases, D-2HG accumulation may lead to cognitive impairment.
- Increased Risk of AML Progression – More aggressive disease course compared to IDH1-negative MDS.
Diagnosis of IDH1-Mutated MDS
A comprehensive diagnostic approach is essential for confirming IDH1 mutations and assessing disease severity.
1. Complete Blood Count (CBC) and Peripheral Blood Smear
- Macrocytic anemia, thrombocytopenia, and dysplastic white cells.
- Pseudo-Pelger-Huët anomaly (abnormally shaped neutrophils).
2. Bone Marrow Biopsy and Aspirate
- Dysplastic erythroid and myeloid precursors.
- Increased myeloblasts (<20% to remain classified as MDS).
3. Molecular and Cytogenetic Testing
- Next-Generation Sequencing (NGS) to detect IDH1 mutations.
- Fluorescence in Situ Hybridization (FISH) to identify additional chromosomal abnormalities.
4. Metabolite Analysis
- Elevated D-2HG levels in serum or bone marrow, indicative of IDH1 activity.
Prognostic Impact of IDH1 Mutations in MDS
The presence of an IDH1 mutation influences prognosis and treatment response:
| Factor | IDH1-Mutated MDS |
|---|---|
| Prognosis | Intermediate to poor |
| AML Progression Risk | Higher than IDH1-negative MDS |
| Response to Therapy | Targetable with IDH1 inhibitors |
| Survival Outcome | Improved with early intervention |
Treatment Approaches for Myelodysplastic Syndrome with IDH1 Mutation
1. Targeted Therapy: IDH1 Inhibitors
- Ivosidenib (AG-120) – FDA-approved IDH1 inhibitor for AML, under investigation for MDS.
- Mechanism – Blocks mutant IDH1, restoring normal differentiation of hematopoietic cells.
- Efficacy – Shown to reduce D-2HG levels, decrease blasts, and improve blood counts.
2. Hypomethylating Agents (HMAs)
- Azacitidine and Decitabine – First-line therapies that improve survival.
- Synergistic Effect – May enhance response when combined with IDH1 inhibitors.
3. Supportive Care
- Blood Transfusions – Alleviate anemia-related symptoms.
- Erythropoiesis-Stimulating Agents (ESAs) – May provide temporary relief in low-risk patients.
- Antibiotic Prophylaxis – Prevents infections due to neutropenia.
4. Hematopoietic Stem Cell Transplantation (HSCT)
- Curative but High-Risk – Considered for younger patients with high-risk disease.
- Timing – Performed in eligible patients following disease stabilization.
Ongoing Research and Future Perspectives
Several clinical trials are exploring new treatment strategies for IDH1-mutated MDS:
- Combination Therapies – IDH1 inhibitors + HMAs to improve remission rates.
- Immunotherapies – Investigating checkpoint inhibitors to enhance immune response.
- Personalized Medicine – Identifying biomarkers to predict treatment response.
Myelodysplastic syndrome with an IDH1 mutation represents a distinct molecular subtype with unique clinical challenges. Advances in targeted therapies, particularly IDH1 inhibitors, have significantly improved treatment options. Early molecular testing and a personalized approach are essential for optimizing patient outcomes.