Non Small Cell Lung Cancer with KRAS G12C Mutation: Non-small cell lung cancer (NSCLC) accounts for approximately 85% of all lung cancer diagnoses globally. It includes subtypes such as adenocarcinoma, squamous cell carcinoma, and large cell carcinoma. Among the many genetic alterations associated with NSCLC, mutations in the KRAS gene have emerged as both common and clinically significant, particularly the G12C substitution.
KRAS Gene and the G12C Mutation in NSCLC
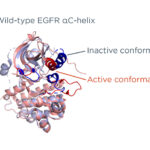
The KRAS (Kirsten rat sarcoma viral oncogene homolog) gene encodes a GTPase involved in cell proliferation and survival signaling. Mutations in this gene result in constitutive activation, driving oncogenesis.
The G12C Substitution Explained
- The G12C mutation involves a glycine-to-cysteine substitution at codon 12 in exon 2 of the KRAS gene.
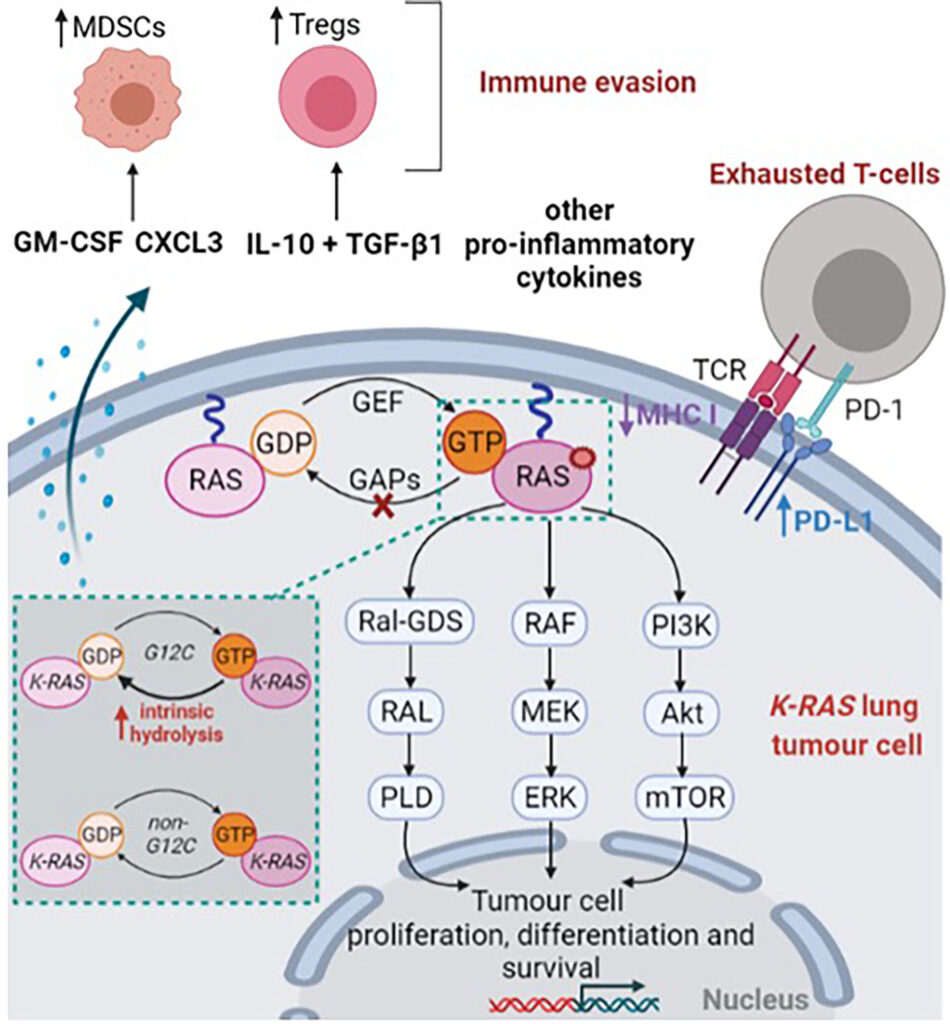
- This alteration locks KRAS in an active GTP-bound state, continuously triggering downstream signaling pathways such as MAPK, PI3K/AKT, and RAL-GDS, promoting cell growth and survival.
Prevalence and Demographics
- The KRAS G12C mutation occurs in approximately 13–15% of NSCLC patients, predominantly in lung adenocarcinoma.
- It is more commonly associated with:
- Current or former smokers
- Western populations compared to Asian demographics
- Patients aged 60 and above
Diagnostic Techniques for KRAS G12C Mutation
Accurate and early identification of KRAS mutations is essential for personalized treatment strategies.
Tissue-Based Molecular Testing
- Performed on tumor samples using:
- Next-generation sequencing (NGS)
- PCR-based assays
- Sanger sequencing
Liquid Biopsy
- Uses circulating tumor DNA (ctDNA) from plasma.
- Allows non-invasive monitoring and mutation detection, especially when tissue is limited.
Companion Diagnostics
- Specific tests like Guardant360 CDx and FoundationOne Liquid CDx are FDA-approved to detect KRAS G12C and guide therapeutic decisions.
Targeted Therapies for KRAS G12C Mutation in NSCLC
Breakthrough with Direct KRAS G12C Inhibitors
Historically considered “undruggable,” the KRAS G12C mutation has recently become a viable therapeutic target.
Sotorasib (AMG 510)
- First FDA-approved KRAS G12C inhibitor (2021)
- Binds irreversibly to the cysteine residue in KRAS G12C
- Shown to improve progression-free survival in pre-treated patients
- Dose: 960 mg orally once daily
- Common side effects: diarrhea, nausea, hepatotoxicity
Adagrasib (MRTX849)
- A second-generation KRAS G12C inhibitor
- Longer half-life, allowing sustained inhibition
- Effective against central nervous system metastases
- Dose: 600 mg orally twice daily
- Side effects include fatigue, QT prolongation, GI toxicity
Efficacy and Clinical Trial Outcomes
CodeBreaK 100 (Sotorasib)
- Objective Response Rate (ORR): ~37%
- Median Progression-Free Survival: 6.8 months
- Median Overall Survival: 12.5 months
KRYSTAL-1 (Adagrasib)
- ORR: ~43%
- Median PFS: 6.5 months
- Active against brain metastases in 33% of evaluated patients
Resistance Mechanisms to KRAS G12C Inhibitors
Despite initial responses, many patients experience acquired resistance.
Resistance Pathways:
- Secondary KRAS mutations (e.g., Y96D)
- Activation of alternate pathways: EGFR, HER2, MET amplification
- Histologic transformation
- Immune microenvironment modulation
To combat resistance, combination therapy is under exploration.
Emerging Combination Therapies and Pipeline Agents
KRAS Inhibitor Combinations
- KRAS G12C inhibitor + SHP2 inhibitor: targets upstream RAS activation
- KRAS inhibitor + MEK inhibitor: dual blockade of the MAPK pathway
- KRAS G12C inhibitor + Immune Checkpoint Inhibitor (ICI): boosts anti-tumor immune response
- KRAS inhibitor + CDK4/6 inhibitors: targets cell cycle progression
Pipeline Candidates
- JAB-21822
- LY3537982
- BI 1823911
These agents are currently in early-stage trials with promising preliminary results.
Role of Immunotherapy in KRAS G12C-Positive NSCLC
- KRAS mutations are often associated with high tumor mutational burden (TMB) and PD-L1 expression.
- Many patients benefit from anti-PD-1/PD-L1 immunotherapies such as pembrolizumab or nivolumab.
- Combination regimens with ICIs and KRAS G12C inhibitors are being evaluated in ongoing clinical trials.
Prognosis and Survival Metrics
- Historically, KRAS mutations were linked to poor prognosis and limited treatment options.
- Introduction of targeted therapies has improved median survival:
- Sotorasib-treated patients: ~12.5 months
- Adagrasib-treated patients: ~14.1 months
- CNS involvement remains a challenge, with adagrasib showing greater intracranial activity than sotorasib.
Clinical Guidelines and Expert Recommendations
- NCCN and ESMO guidelines endorse routine testing for KRAS mutations in metastatic NSCLC.
- For KRAS G12C-positive patients:
- First-line treatment: chemotherapy +/- immunotherapy (if not previously treated)
- Second-line: KRAS G12C inhibitors post-progression
- Clinical trials are encouraged for patients who develop resistance
The discovery and therapeutic targeting of the KRAS G12C mutation in non-small cell lung cancer represent a milestone in precision oncology. With the development of sotorasib and adagrasib, patients with previously limited options now benefit from mutation-specific treatments. As research advances, combinatorial approaches and novel agents hold promise for overcoming resistance and improving survival outcomes.