Non Small Cell Lung Cancer with EGFR Exon 21 L858R: Non small cell lung cancer (NSCLC) accounts for approximately 85% of all lung cancer cases. It encompasses a group of lung cancers, including adenocarcinoma, squamous cell carcinoma, and large cell carcinoma. A significant number of NSCLC patients present with specific genetic alterations, among which mutations in the epidermal growth factor receptor (EGFR) gene are clinically crucial for targeted therapy.
EGFR Exon 21 L858R Substitution Mutation: A Key Oncogenic Driver
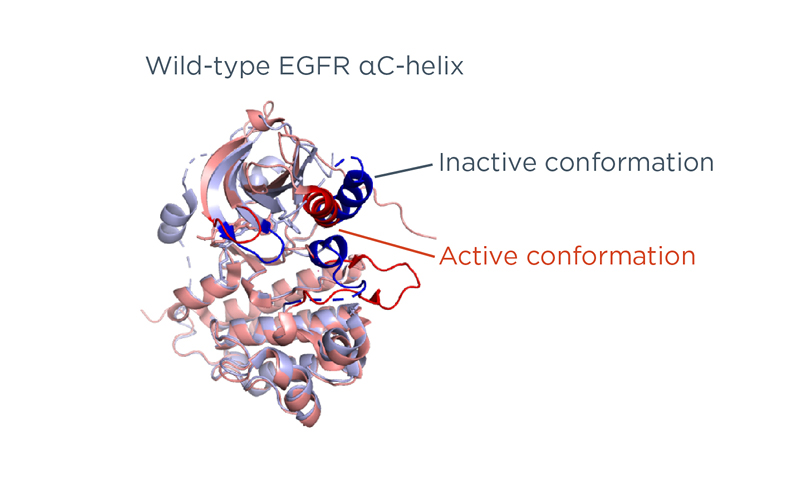
The L858R mutation in exon 21 of the EGFR gene is a missense mutation, substituting leucine (L) with arginine (R) at position 858. This alteration leads to constitutive activation of the EGFR tyrosine kinase domain, resulting in uncontrolled cellular proliferation and survival. Alongside exon 19 deletions, the L858R mutation is one of the most prevalent EGFR mutations in NSCLC, together comprising approximately 90% of sensitizing EGFR mutations.
Diagnostic Approaches for L858R Mutation in NSCLC
Tissue Biopsy and Molecular Testing
- Formalin-fixed paraffin-embedded (FFPE) tumor tissue is the gold standard.
- Techniques include:
- Real-time PCR
- Next-generation sequencing (NGS)
- Digital droplet PCR (ddPCR)
- Sanger sequencing
Liquid Biopsy
- Non-invasive option using circulating tumor DNA (ctDNA).
- Allows real-time monitoring of mutation status and resistance mechanisms.
Companion Diagnostics for Non Small Cell Lung Cancer with EGFR Exon 21 L858R
FDA-approved companion diagnostics are required before prescribing EGFR tyrosine kinase inhibitors (TKIs). These diagnostics ensure mutation-specific targeting.
Targeted Therapy Options for L858R-Positive NSCLC
First-Generation EGFR TKIs
- Erlotinib and Gefitinib:
- Reversible inhibitors
- Approved for frontline treatment in EGFR-mutated NSCLC
- Median progression-free survival (PFS): 9–11 months
Second-Generation EGFR TKIs
- Afatinib and Dacomitinib:
- Irreversible pan-HER inhibitors
- Broader inhibitory spectrum
- Associated with longer PFS but increased toxicity
Third-Generation EGFR TKIs
- Osimertinib:
- Irreversibly binds to both sensitizing mutations (like L858R) and T790M resistance mutations
- Crosses the blood-brain barrier, making it effective for CNS metastases
- FLAURA Trial: Median PFS of 18.9 months vs 10.2 months for first-gen TKIs
Resistance Mechanisms and Disease Progression
Despite initial efficacy, most patients eventually experience disease progression due to acquired resistance.
Common Resistance Mechanisms:
- EGFR T790M mutation: Emerges in 50–60% of cases post first-gen TKI treatment
- MET amplification
- HER2 amplification
- Histologic transformation to small cell lung cancer
- EMT (Epithelial-to-Mesenchymal Transition)
Ongoing monitoring using liquid biopsies and rebiopsies is essential to detect resistance pathways and guide subsequent therapy.
Emerging Therapies and Ongoing Clinical Trials
Combination Strategies
- Osimertinib + MET inhibitors (e.g., Savolitinib)
- EGFR TKIs + Anti-angiogenics (e.g., Osimertinib + Bevacizumab)
- EGFR TKIs + Immune Checkpoint Inhibitors: Trials ongoing despite mixed results due to increased pneumonitis risk
Novel Agents Under Investigation
- Amivantamab: EGFR-MET bispecific antibody
- Lazertinib: Third-generation TKI with promising CNS penetration
These agents are particularly relevant for patients with exon 21 L858R mutation who relapse on current EGFR-TKI therapy.
Prognosis and Survival Outcomes
Patients harboring the EGFR L858R mutation generally have better initial responses to EGFR-TKIs than those without EGFR mutations. However, several studies indicate that:
- Exon 19 deletions may yield better survival outcomes than L858R mutations
- L858R mutation carriers may develop resistance more rapidly
- Median overall survival (OS) with Osimertinib in L858R-positive patients ranges from 32–38 months
Clinical Guidelines and Recommendations
- NCCN and ESMO recommend EGFR mutation testing in all NSCLC adenocarcinoma patients, particularly non-smokers.
- First-line treatment for EGFR L858R mutation-positive NSCLC is Osimertinib due to superior efficacy and CNS activity.
- Upon progression, rebiopsy and molecular profiling are recommended to determine resistance mechanisms and tailor second-line therapy.
The EGFR exon 21 L858R substitution mutation represents a pivotal molecular target in the management of NSCLC. Advances in precision diagnostics and the development of EGFR-targeted therapies have transformed the treatment landscape, enabling improved patient outcomes. Continuous research into resistance mechanisms and novel therapeutic combinations is essential to sustain and enhance therapeutic success in this subgroup.