Non small cell lung cancer with braf v600e mutation: Non-small cell lung cancer (NSCLC) represents the majority of lung cancer diagnoses worldwide, with several molecular alterations guiding treatment decisions. Among these, the BRAF V600E mutation stands out as a significant oncogenic driver. Though relatively rare, occurring in approximately 1–4% of NSCLC cases, this mutation leads to constitutive activation of the MAPK/ERK pathway, fueling uncontrolled tumor growth.
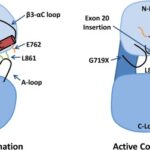
Molecular Biology of BRAF V600E in NSCLC
The BRAF gene encodes a serine/threonine kinase that plays a pivotal role in the RAS-RAF-MEK-ERK signaling cascade. The V600E mutation results from a substitution of valine (V) with glutamic acid (E) at codon 600, triggering continuous kinase activity and promoting cell proliferation independent of external growth signals.
This mutation-driven signaling enhances tumor aggressiveness and influences therapeutic responsiveness.
Epidemiology and Clinical Features
- Incidence: BRAF mutations occur in 1–4% of NSCLC patients; among these, V600E accounts for ~50% of BRAF alterations.
- Histology: Predominantly associated with lung adenocarcinoma.
- Demographics: Unlike other driver mutations (e.g., EGFR), BRAF V600E mutations can be found in both smokers and non-smokers.
- Gender Distribution: A slight female predominance has been observed in some cohorts.
Diagnostic Approach for BRAF-Positive NSCLC
Tissue Sampling and Histopathology
- Lung biopsy (via bronchoscopy, CT-guided, or surgical resection)
- Histologic classification—typically adenocarcinoma with variable differentiation
Molecular Testing
Accurate identification of BRAF V600E mutation is essential to initiate targeted therapy.
- Next-generation sequencing (NGS): Comprehensive detection of multiple mutations including BRAF
- PCR-based assays: Mutation-specific testing
- Immunohistochemistry (IHC): Limited utility, often used in adjunctive capacity
Routine molecular profiling of all advanced NSCLC cases is recommended to identify actionable mutations such as BRAF.
Targeted Therapy for NSCLC with BRAF V600E Mutation
Approved Targeted Agents
The FDA-approved treatment for BRAF V600E-positive NSCLC includes a dual inhibition approach using:
- Dabrafenib (BRAF inhibitor)
- Trametinib (MEK inhibitor)
Mechanism of Action
- Dabrafenib blocks the mutant BRAF protein, preventing MAPK pathway activation.
- Trametinib inhibits downstream MEK, ensuring sustained pathway suppression.
This combination prevents feedback reactivation and delays resistance.
Clinical Trial Evidence
The BRF113928 trial demonstrated:
- Overall response rate (ORR): ~64% in previously treated patients
- Median progression-free survival (PFS): ~10.9 months
- Median overall survival (OS): ~24.6 months
Both previously treated and treatment-naïve patients showed durable responses with the combination therapy.
Treatment Algorithm for BRAF V600E-Mutated NSCLC
| Disease Stage | Recommended Treatment |
|---|---|
| Early Stage (I–II) | Surgery ± adjuvant chemotherapy |
| Locally Advanced (III) | Chemoradiation ± consolidation immunotherapy |
| Advanced/Metastatic (IV) | Dabrafenib + Trametinib (1st line or post-chemotherapy) |
For patients who progress on targeted therapy, standard chemotherapy or immunotherapy may be considered, although efficacy is generally reduced.
Side Effects and Management
Common Adverse Effects of Dabrafenib + Trametinib
- Pyrexia (fever)
- Fatigue
- Nausea
- Diarrhea
- Skin rash
- Photosensitivity
- Liver enzyme elevation
Management involves dose modifications, antipyretics, and temporary treatment interruptions.
Comparison with Other Oncogenic Drivers
| Mutation | Targeted Therapy | ORR (%) | PFS (months) |
|---|---|---|---|
| EGFR | Osimertinib | ~70 | ~18.9 |
| ALK | Alectinib | ~80 | ~34.8 |
| BRAF V600E | Dabrafenib + Trametinib | ~64 | ~10.9 |
| ROS1 | Crizotinib | ~72 | ~19.3 |
While response rates in BRAF-mutant NSCLC are slightly lower than other driver mutations, targeted therapy still offers a significant advantage over chemotherapy.
Immunotherapy and BRAF V600E-Positive NSCLC
Patients with BRAF V600E mutations may have lower tumor mutational burden (TMB) and PD-L1 expression, leading to variable response to immune checkpoint inhibitors (ICIs). Immunotherapy may be used:
- After progression on targeted therapy
- In combination with chemotherapy (if targetable mutation is unknown)
- If PD-L1 expression is ≥50% and patient is ineligible for targeted therapy
Monitoring, Resistance, and Disease Progression
Monitoring Strategy
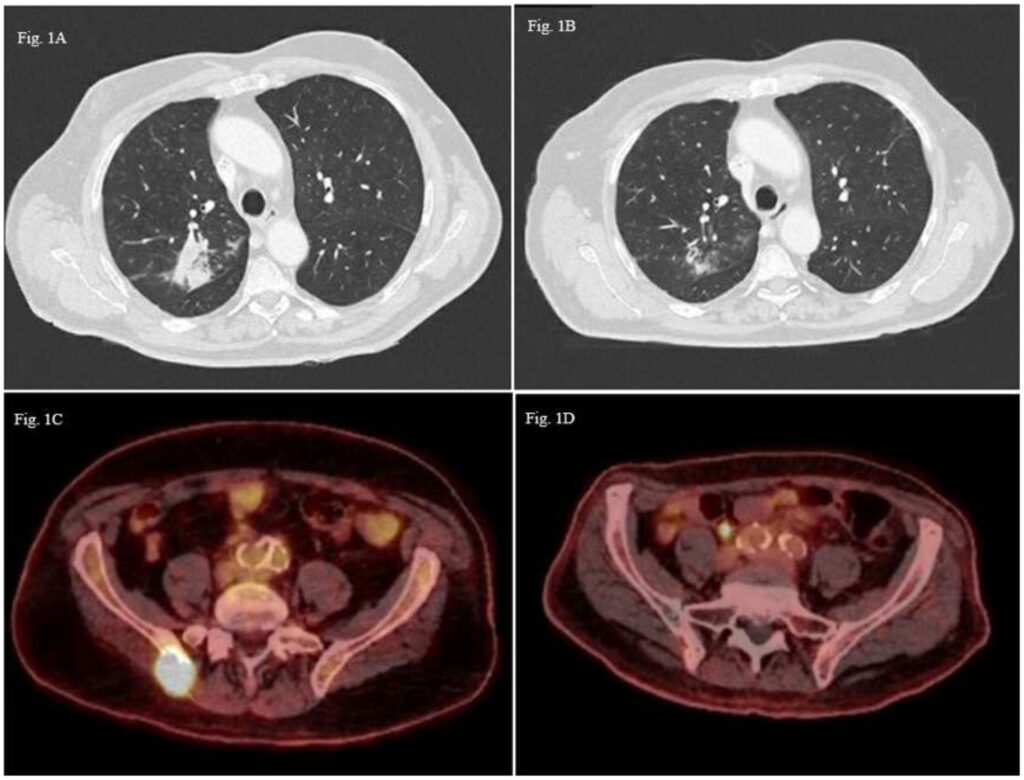
- Imaging: Every 2–3 months to assess response
- CT or PET-CT scans
- Tumor markers (optional, non-specific)
Resistance Mechanisms
Resistance to dabrafenib/trametinib may emerge via:
- Secondary mutations in MEK1 or other MAPK components
- Alternative pathway activation (e.g., PI3K/AKT)
- Histologic transformation
Rebiopsy and repeat molecular profiling can guide second-line strategies.
Future Directions and Ongoing Research
- Triplet therapies combining BRAF, MEK, and immune checkpoint inhibitors
- Selective next-generation BRAF inhibitors
- Combination with anti-angiogenic agents
- Liquid biopsy advancements for non-invasive mutation detection
Clinical trials continue to explore avenues for overcoming resistance and extending survival in BRAF V600E-mutated NSCLC.
Non-small cell lung cancer harboring the BRAF V600E mutation is a biologically distinct subset that benefits significantly from molecularly targeted therapy. The combination of dabrafenib and trametinib has emerged as the standard of care, offering improved outcomes compared to traditional chemotherapy. As research evolves, we anticipate the development of newer agents and combination regimens to further improve patient prognosis and quality of life. Timely diagnosis, genetic profiling, and personalized treatment planning remain the cornerstones of effective care in this rare but impactful NSCLC subtype.