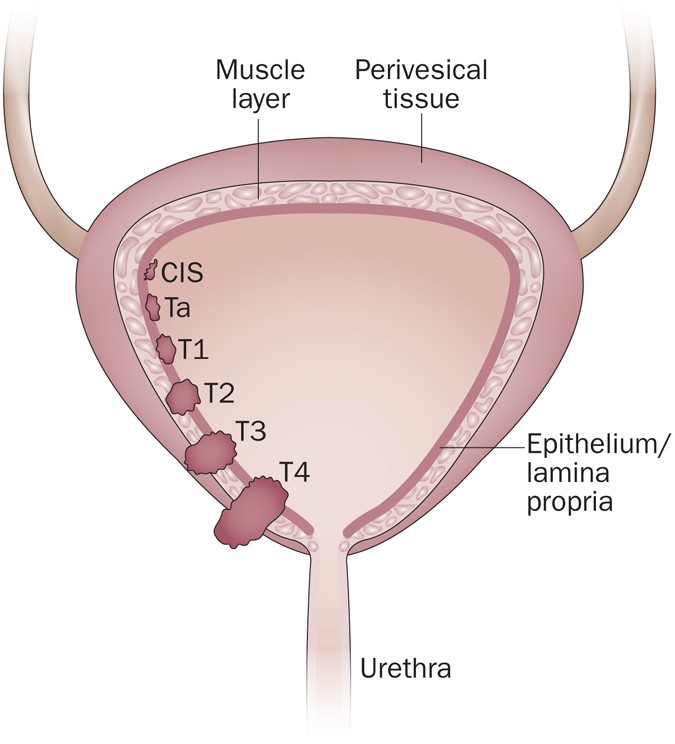
Non-muscle invasive bladder cancer (NMIBC) comprises a heterogeneous group of urothelial malignancies confined to the mucosa (Ta, carcinoma in situ) or submucosa (T1) of the bladder wall, without invasion into the muscularis propria. Among these, carcinoma in situ (CIS) represents a high-grade, flat, and aggressive form of NMIBC, often associated with a high risk of progression and recurrence.
Understanding Carcinoma in Situ of the Bladder
Definition and Histopathology
Carcinoma in situ (CIS) is defined as a flat, high-grade, non-invasive urothelial carcinoma that may occur in isolation or concurrently with papillary tumors (Ta, T1). Unlike papillary lesions, CIS does not protrude into the bladder lumen, making it more challenging to detect visually and radiologically.
CIS is histologically characterized by:
- Loss of cellular polarity
- Enlarged, pleomorphic nuclei
- Prominent nucleoli
- High mitotic activity
- Absence of stromal invasion
Clinical Presentation
Patients with CIS often present with:
- Irritative voiding symptoms (urgency, frequency, dysuria)
- Microscopic or gross hematuria
- May be asymptomatic and detected during surveillance cystoscopy
CIS is strongly associated with multifocality, positive urinary cytology, and field cancerization, increasing the risk of recurrence and progression.
Diagnostic Approach to NMIBC with CIS
Initial Evaluation
Diagnosis involves a multi-modal approach combining:
- Cystoscopy: White-light cystoscopy may miss flat lesions; thus, enhanced techniques are recommended.
- Fluorescence Cystoscopy (Blue Light Cystoscopy): Improves detection of CIS by highlighting abnormal areas using hexaminolevulinate (HAL).
- Urine Cytology: High sensitivity for high-grade lesions such as CIS.
- Transurethral Resection of Bladder Tumor (TURBT): Crucial for histologic confirmation and staging.
Bladder Cancer Staging and Risk Stratification
Pathological Staging
- CIS (Tis): High-grade, non-invasive, flat lesion
- Often coexists with Ta (non-invasive papillary) or T1 (lamina propria invasive) disease
Risk Stratification (per EAU/AUA guidelines)
Patients with CIS fall into the high-risk category due to:
- High-grade nature
- High recurrence and progression rates
- Potential for upper tract involvement
Management of Non-Muscle Invasive Bladder Cancer with CIS
Standard Initial Treatment: Intravesical BCG Therapy
Bacillus Calmette-Guérin (BCG) is the gold standard for high-risk NMIBC, including CIS. Its immunomodulatory effects induce local immune responses that eradicate residual tumor cells.
BCG Treatment Protocol:
- Induction phase: Weekly instillations for 6 weeks
- Maintenance phase: 3-week cycles at 3, 6, 12, 18, 24, 30, and 36 months (per SWOG protocol)
Response Evaluation
- Urine cytology and cystoscopy at 3 months post-induction
- Complete responders continue with maintenance
- Non-responders require prompt reassessment
BCG-Refractory or Unresponsive CIS
Defined as persistent high-grade disease at 6 months despite adequate BCG. Management options include:
- Radical cystectomy (standard of care for fit patients)
- Intravesical alternatives:
- Valrubicin (FDA-approved but low efficacy)
- Clinical trials with checkpoint inhibitors (e.g., nivolumab, atezolizumab)
- Gene therapy agents (e.g., nadofaragene firadenovec)
Radical Cystectomy: Role in CIS Management
Indications
- BCG-unresponsive CIS
- Multifocal or extensive CIS
- Rapid progression or intolerance to intravesical therapy
Outcomes
Early cystectomy offers:
- 90% 5-year cancer-specific survival
- Reduced risk of muscle-invasive transformation
Bladder-sparing approaches should be reserved for selected patients with limited, unifocal CIS and close surveillance.
Surveillance and Recurrence Monitoring
Post-Treatment Surveillance Schedule
| Time Frame | Investigations |
|---|---|
| Every 3 months | Cystoscopy + Urine Cytology |
| Every 6 months | Upper tract imaging (1st year) |
| Annually | Continued cystoscopic exams |
Early detection of recurrence, particularly high-grade lesions, is crucial to prevent disease progression.
Prognosis and Long-Term Outcomes
Recurrence and Progression Rates
- Recurrence: Up to 60% within 1–2 years
- Progression to muscle-invasive disease: 10–20% over 5 years
- Higher risk in patients with:
- Concomitant T1 disease
- Multifocal CIS
- Incomplete BCG therapy
Prognostic Indicators
- Response to initial BCG therapy
- Negative urinary cytology
- Extent and location of CIS
Emerging Therapies and Research
Checkpoint Inhibitors and Immunotherapy
Immune checkpoint blockade has shown promise in BCG-unresponsive CIS, with trials evaluating:
- Atezolizumab
- Nivolumab
- Pembrolizumab (KEYNOTE-057 trial)
Gene Therapy
- Nadofaragene firadenovec: Delivers interferon-alpha via adenoviral vector
- Achieved ~53% complete response at 3 months in CIS patients
Targeted Drug Delivery Systems
- Electromotive drug administration (EMDA)
- Hyperthermic intravesical chemotherapy (HIVEC)
These novel approaches aim to enhance drug penetration and efficacy, especially in difficult-to-treat CIS.
Non-muscle invasive bladder cancer with carcinoma in situ represents a high-risk yet potentially curable entity when diagnosed early and treated aggressively. The cornerstone of management remains BCG immunotherapy, with close surveillance and timely escalation to radical surgery in unresponsive cases. Ongoing advancements in immunotherapy and gene-based treatments are poised to redefine the therapeutic paradigm and offer renewed hope for patients with BCG-refractory CIS.