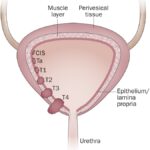
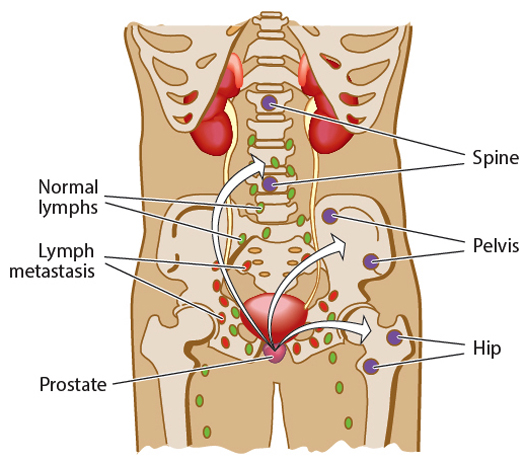
Non-metastatic castration-resistant prostate cancer (nmCRPC) is a clinically distinct phase of prostate cancer characterized by biochemical progression despite androgen deprivation therapy (ADT), in the absence of detectable distant metastases on conventional imaging. Patients typically present with a rising prostate-specific antigen (PSA) level while maintaining castrate levels of testosterone (<50 ng/dL).
This condition demands proactive therapeutic intervention due to the high risk of progression to metastatic disease, especially in those with rapid PSA doubling times (≤10 months).
Pathophysiology and Progression
The evolution of nmCRPC results from adaptive resistance to long-term androgen suppression, where tumor cells continue to proliferate through androgen receptor (AR) pathway activation, mutations, or bypass mechanisms.
Key mechanisms include:
- AR gene amplification or mutations
- Upregulation of co-activators
- Ligand-independent AR activation
- Intratumoral androgen synthesis
Clinical Characteristics and Diagnostic Criteria
Essential Criteria for Diagnosis
- Castration-level testosterone (<50 ng/dL)
- Rising PSA on three consecutive occasions at least one week apart
- No radiographic evidence of metastasis (CT, MRI, or bone scan)
Patients often remain asymptomatic, making regular PSA monitoring critical for early detection.
Risk Stratification
The most significant predictor of metastasis is PSA doubling time:
- ≤10 months: High-risk
- >10 months: Low-risk
This stratification aids in selecting patients who benefit from early therapeutic escalation.
Treatment Landscape for nmCRPC
Continuing Androgen Deprivation Therapy (ADT)
All patients must continue ADT indefinitely to maintain castrate testosterone levels. ADT alone, however, is insufficient to halt progression in high-risk nmCRPC.
Second-Generation AR Inhibitors
New-generation androgen receptor signaling inhibitors (ARSIs) have demonstrated significant metastasis-free survival (MFS) benefits in nmCRPC.
1. Apalutamide (SPARTAN Trial)
- MFS improvement: 40.5 months vs. 16.2 months (placebo)
- PSA doubling time ≤10 months subgroup benefited the most
- Adverse effects: fatigue, rash, hypothyroidism
2. Enzalutamide (PROSPER Trial)
- MFS improvement: 36.6 months vs. 14.7 months
- Also showed delayed time to first use of chemotherapy
- Side effects include hypertension, falls, cognitive effects
3. Darolutamide (ARAMIS Trial)
- MFS improvement: 40.4 months vs. 18.4 months
- Favorable safety profile, minimal CNS penetration
- Reduced incidence of fatigue, falls, and cognitive issues
These agents are FDA-approved and form the standard of care in patients with nmCRPC and rapid PSA doubling.
Comparative Summary of ARSIs
| Agent | MFS (months) | Key Benefit | Common Side Effects |
|---|---|---|---|
| Apalutamide | 40.5 | Delayed metastasis, OS benefit | Rash, hypothyroidism, fatigue |
| Enzalutamide | 36.6 | Time to chemo delayed | Hypertension, fatigue, memory |
| Darolutamide | 40.4 | Best tolerability | Low CNS-related AEs, fatigue |
Monitoring and Imaging in nmCRPC
PSA Surveillance
- PSA every 3–6 months
- PSA doubling time is recalculated to assess risk progression
Imaging Modalities
- CT and bone scans remain standard
- PSMA PET imaging offers higher sensitivity, but its role in nmCRPC is evolving, as detection of early metastases may reclassify patients as mCRPC
Management of Adverse Events
Treatment-related adverse effects require vigilant monitoring:
- Fatigue management: physical activity, nutritional support
- Bone health: calcium, vitamin D, bisphosphonates or denosumab
- Cardiovascular monitoring: baseline ECG, BP control
- Falls and cognitive safety: particularly relevant for elderly populations
Dose modifications and supportive care strategies are essential to maintain therapy adherence.
Emerging Therapies and Research Directions
Ongoing studies are evaluating:
- Combination strategies: ARSI + radiopharmaceuticals
- Immunotherapy: exploring checkpoint inhibitors
- Novel biomarkers: circulating tumor cells, genomic alterations for personalized treatment
- Use of PSMA PET: to guide earlier systemic therapy in molecularly metastatic disease
Prognosis and Survival Outcomes
Early intervention in high-risk nmCRPC with AR inhibitors significantly delays metastasis, improves quality of life, and extends overall survival. Patients treated with ARSIs can often maintain normal daily activities, particularly when toxicity is managed appropriately.
Non-metastatic castration-resistant prostate cancer represents a pivotal therapeutic window in the management of advanced prostate cancer. The incorporation of second-generation AR inhibitors has transformed the prognosis of high-risk nmCRPC patients, offering meaningful extensions in metastasis-free and overall survival. With continual advancements in imaging, molecular diagnostics, and targeted therapies, the future of nmCRPC management is increasingly personalized and precise.