Myeloid blast phase Philadelphia chromosome-positive chronic myelocytic leukemia (CML-BP) represents the most aggressive stage of CML. It is characterized by an excess of myeloid blasts (≥20%) in the blood or bone marrow, leading to rapid disease progression and poor prognosis. The transition from chronic phase (CP) to blast phase (BP) is often driven by additional genetic abnormalities, resistance to tyrosine kinase inhibitors (TKIs), and clonal evolution. Understanding the molecular mechanisms behind CML blast crisis is crucial for developing effective treatment strategies.
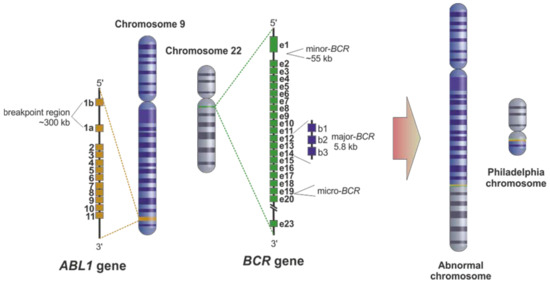
Pathophysiology of Myeloid Blast Phase CML
CML originates from a genetic translocation known as the Philadelphia chromosome (Ph+), t(9;22)(q34;q11), which results in the formation of the BCR-ABL1 fusion gene. This fusion encodes a constitutively active tyrosine kinase, promoting uncontrolled myeloid proliferation.
As the disease progresses into the blast phase, additional mutations and chromosomal aberrations accumulate, leading to therapy resistance and an acute leukemia-like phenotype. Common secondary mutations include:
- TP53 loss – Enhances genomic instability.
- RUNX1 mutations – Disrupt normal hematopoiesis.
- ASXL1 mutations – Drive clonal evolution.
- NRAS/KRAS mutations – Activate oncogenic signaling pathways.
Causes and Risk Factors
Several factors contribute to the transformation of CML chronic phase into blast crisis:
- Acquisition of Additional Genetic Mutations – Clonal evolution leads to therapy-resistant leukemic clones.
- Failure of Tyrosine Kinase Inhibitor (TKI) Therapy – Resistance mechanisms include BCR-ABL1 kinase domain mutations (e.g., T315I mutation).
- Epigenetic Dysregulation – Aberrant DNA methylation and histone modifications drive leukemic transformation.
- Inflammatory Microenvironment – Chronic inflammation promotes blast proliferation and resistance.
Symptoms of Myeloid Blast Phase CML
The clinical presentation of CML in blast phase resembles acute leukemia and includes:
- Severe Fatigue and Weakness – Due to bone marrow failure.
- Uncontrolled Bleeding and Bruising – Resulting from thrombocytopenia.
- Persistent Fevers and Night Sweats – Indicative of systemic inflammation.
- Enlarged Spleen (Splenomegaly) – Causing abdominal discomfort and early satiety.
- Bone Pain and Joint Discomfort – Due to leukemic infiltration.
Diagnosis of Myeloid Blast Phase CML
1. Blood and Bone Marrow Analysis
- Complete Blood Count (CBC) – Shows leukocytosis with increased blast cells.
- Peripheral Blood Smear – Identifies circulating myeloid blasts.
- Bone Marrow Biopsy – Confirms ≥20% blast cells and fibrosis.
2. Cytogenetic and Molecular Testing
- Fluorescence In Situ Hybridization (FISH) – Detects the BCR-ABL1 fusion gene.
- Quantitative Polymerase Chain Reaction (qPCR) – Measures BCR-ABL1 transcript levels.
- Next-Generation Sequencing (NGS) – Identifies additional mutations driving blast transformation.
3. Flow Cytometry
- Differentiates myeloid vs. lymphoid blast phase based on surface markers (e.g., CD34, CD117, MPO for myeloid lineage).
Prognosis and Risk Stratification
Blast phase CML carries a poor prognosis, with a median survival of 3 to 6 months without treatment. Prognostic indicators include:
| Risk Factor | Prognostic Impact |
|---|---|
| BCR-ABL1 Kinase Domain Mutations | Reduced TKI efficacy |
| High Blast Count (>50%) | Increased mortality risk |
| TP53 or RUNX1 Mutations | Poor response to therapy |
| Failure of Second-Generation TKIs | Increased risk of relapse |
Treatment Strategies for Myeloid Blast Phase CML
1. Tyrosine Kinase Inhibitor (TKI) Therapy
Despite resistance, TKIs remain the first-line treatment:
- Ponatinib – Effective for T315I mutation-positive cases.
- Dasatinib or Nilotinib – Used if prior TKIs fail.
2. Chemotherapy-Based Approaches
Blast phase CML often requires AML-like induction chemotherapy:
- Cytarabine + Anthracyclines (7+3 regimen) – Standard induction therapy.
- Hypomethylating Agents (Azacitidine, Decitabine) – For elderly or frail patients.
3. Allogeneic Hematopoietic Stem Cell Transplantation (HSCT)
The only curative option, recommended for eligible patients after remission induction.
4. Experimental Therapies and Clinical Trials
- BCL-2 Inhibitors (Venetoclax) – Target apoptosis pathways.
- FLT3 and IDH Inhibitors – Investigated for targeted therapy.
- CAR-T Therapy and Immunotherapies – Emerging options for treatment-resistant cases.
Future Perspectives and Research
Current research focuses on combination therapies integrating TKIs with novel agents to overcome resistance. Personalized medicine approaches, guided by genomic and transcriptomic profiling, hold promise for improving outcomes in blast phase CML.
Myeloid blast phase Philadelphia chromosome-positive chronic myelocytic leukemia represents a major therapeutic challenge. Early identification of high-risk patients, mutation-directed therapy, and stem cell transplantation are crucial for improving survival rates. As research progresses, novel targeted treatments may provide new hope for patients with advanced CML.