Myelofibrosis (MF) is a rare but serious myeloproliferative neoplasm (MPN) characterized by abnormal fibrotic tissue accumulation in the bone marrow. This disrupts normal blood cell production, leading to anemia, an enlarged spleen (splenomegaly), and a high risk of transformation into acute myeloid leukemia (AML). Advances in targeted therapies have improved patient outcomes, yet myelofibrosis remains a challenging condition requiring personalized treatment strategies.
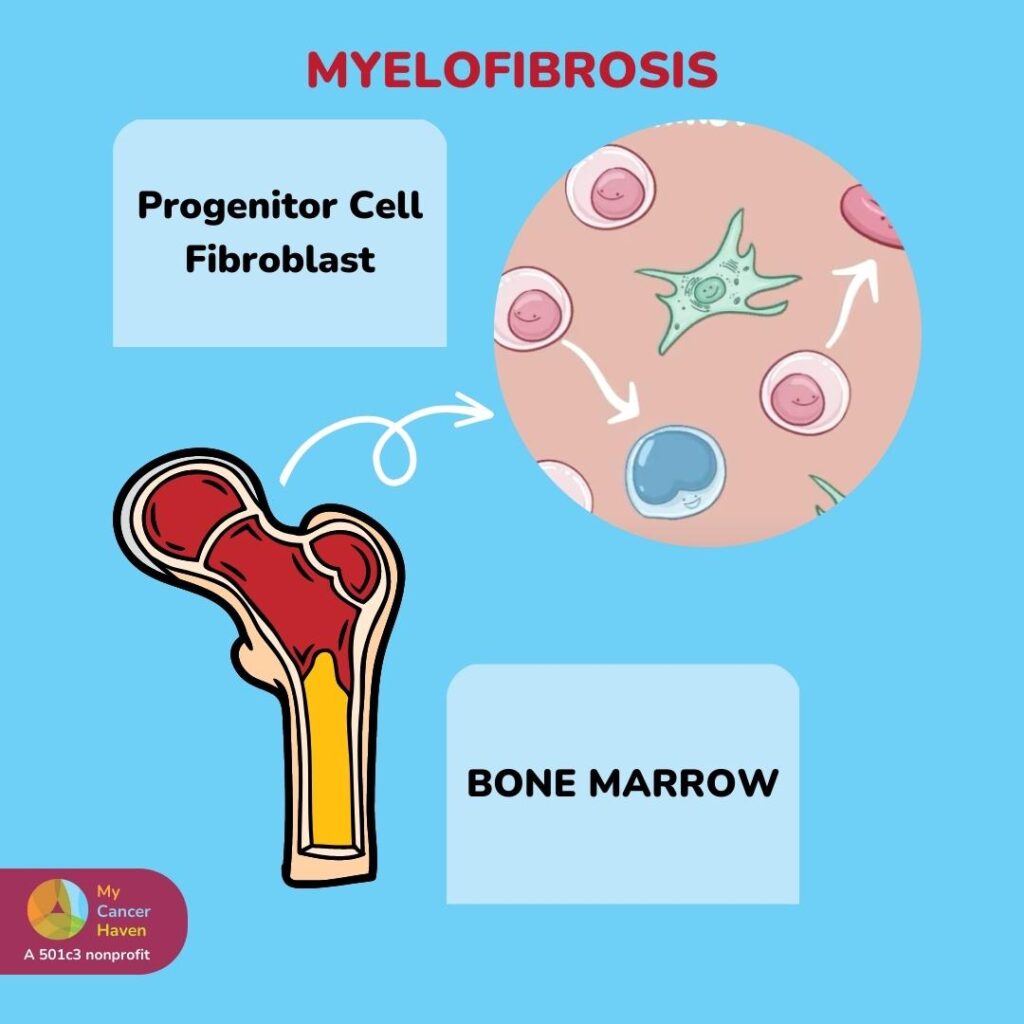
Pathophysiology of Myelofibrosis
Myelofibrosis results from mutations in key genes involved in hematopoiesis, particularly JAK2, CALR, and MPL. These mutations cause abnormal signaling in hematopoietic stem cells, leading to excessive production of fibrogenic cytokines such as transforming growth factor-beta (TGF-β) and platelet-derived growth factor (PDGF), which stimulate fibroblast proliferation in the bone marrow.
This fibrotic process disrupts normal blood formation, forcing blood cell production to shift to organs such as the spleen and liver, a condition known as extramedullary hematopoiesis (EMH).
Causes and Risk Factors
Although the exact cause of myelofibrosis remains unclear, several genetic and environmental factors contribute to its development:
- Genetic Mutations – JAK2 (found in ~60% of cases), CALR (~20-25%), and MPL (~5-10%) mutations drive abnormal blood cell proliferation.
- Age and Gender – Most commonly diagnosed in individuals over 50, with a slight male predominance.
- Exposure to Toxins – Prior exposure to radiation, benzene, or industrial chemicals increases the risk.
- Secondary Myelofibrosis – Can develop from other MPNs such as polycythemia vera (PV) or essential thrombocythemia (ET).
Symptoms of Myelofibrosis
Myelofibrosis symptoms vary based on disease severity, but common clinical manifestations include:
- Fatigue and Weakness – Due to anemia from inadequate red blood cell production.
- Splenomegaly (Enlarged Spleen) – Causes abdominal pain, early satiety, and weight loss.
- Bone Pain – Resulting from marrow fibrosis and osteosclerosis.
- Night Sweats and Fever – Systemic symptoms linked to inflammatory cytokine release.
- Easy Bruising and Bleeding – Due to platelet dysfunction.
- Gout or High Uric Acid Levels – A consequence of increased cell turnover.
Diagnosis of Myelofibrosis
A comprehensive diagnostic approach is crucial to differentiate myelofibrosis from other MPNs.
1. Blood Tests
- Complete Blood Count (CBC) – Shows anemia, variable white blood cell counts, and abnormal platelet levels.
- Peripheral Blood Smear – Reveals tear-drop-shaped red blood cells (dacrocytes) and immature granulocytes.
- Lactate Dehydrogenase (LDH) – Elevated due to high cell turnover.
2. Bone Marrow Biopsy
- Identifies reticulin or collagen fibrosis, abnormal megakaryocytes, and reduced hematopoietic precursors.
3. Genetic Testing
- JAK2, CALR, and MPL mutation analysis aids in diagnosis and risk stratification.
4. Imaging Studies
- Abdominal Ultrasound or MRI – Detects splenomegaly and extramedullary hematopoiesis.
Prognosis and Risk Stratification
Several scoring systems help assess myelofibrosis prognosis, including DIPSS (Dynamic International Prognostic Scoring System). Factors influencing survival include:
| Risk Factor | Prognostic Impact |
|---|---|
| Age >65 years | Shortened survival |
| Anemia (Hb <10 g/dL) | Poor prognosis |
| Leukocytosis (>25,000/µL) | High-risk category |
| Circulating Blasts (>1%) | Increased risk of AML transformation |
| JAK2, CALR, MPL Mutations | Affects response to therapy |
Treatment Strategies for Myelofibrosis
1. JAK Inhibitors: Targeted Therapy
JAK inhibitors are first-line treatment options for symptomatic myelofibrosis.
- Ruxolitinib (Jakafi®) – FDA-approved JAK1/2 inhibitor, reduces spleen size and improves survival.
- Fedratinib (Inrebic®) – Used in ruxolitinib-resistant cases, effective against severe splenomegaly.
- Pacritinib and Momelotinib – Options for patients with severe anemia.
2. Supportive Care
- Blood Transfusions – Manage anemia-related symptoms.
- Erythropoiesis-Stimulating Agents (ESAs) – For low erythropoietin levels.
- Iron Chelation Therapy – Prevents iron overload from transfusions.
3. Allogeneic Stem Cell Transplantation (HSCT)
- Only curative option but associated with high risks.
- Recommended for high-risk patients under 65 with a compatible donor.
4. Experimental Therapies and Clinical Trials
- BET Inhibitors – Target epigenetic regulators to reduce fibrosis.
- Telomerase Inhibitors (Imetelstat) – Show promise in disease modification.
- Combination Therapies – JAK inhibitors + novel agents under investigation.
Outlook and Future Research
Research efforts continue to refine personalized treatment approaches, improve survival rates, and develop therapies to reverse fibrosis. New drug combinations and immunotherapies hold promise for transforming myelofibrosis management in the coming years.
Myelofibrosis remains a complex hematologic disorder with a variable prognosis. Early diagnosis, genetic risk assessment, and targeted therapies significantly improve patient outcomes. Ongoing advancements in treatment strategies offer hope for more effective management and potential disease-modifying approaches.