Myelodysplastic syndrome with 5q deletion (del(5q) MDS) is a distinct subtype of myelodysplastic syndromes (MDS) characterized by a deletion in the long arm of chromosome 5. This abnormality affects bone marrow function, leading to anemia and other hematologic abnormalities. Del(5q) MDS is often associated with a favorable prognosis compared to other MDS subtypes and responds well to specific treatments, such as lenalidomide.
Pathophysiology and Genetic Basis
The deletion of chromosome 5q results in the loss of essential genes involved in hematopoiesis, leading to ineffective blood cell production and bone marrow dysplasia. Key genes affected include:
- RPS14 – Implicated in erythroid maturation; loss leads to anemia.
- CSNK1A1 – Involved in stem cell regulation; a target of lenalidomide.
- EGR1 and APC – Affect cellular proliferation and differentiation.
Causes and Risk Factors
The exact cause of del(5q) MDS remains unclear, but several risk factors increase its likelihood:
- Age – Primarily affects individuals over 60.
- Previous Chemotherapy or Radiation – Alkylating agents and topoisomerase inhibitors increase risk.
- Environmental Exposure – Benzene, pesticides, and heavy metals are linked to MDS.
- Genetic Susceptibility – Some inherited conditions predispose individuals to chromosomal abnormalities.
Symptoms of del(5q) MDS
Del(5q) MDS primarily presents with symptoms related to anemia:
- Fatigue and Weakness – Reduced red blood cell production leads to low oxygen levels.
- Pallor and Shortness of Breath – Common signs of chronic anemia.
- Mild Leukopenia and Thrombocytosis – Unlike other MDS types, platelet counts may be elevated.
- Rarely, Progression to Acute Myeloid Leukemia (AML) – Though less common, monitoring is essential.
Diagnosis of Myelodysplastic Syndrome with 5q Deletion
A combination of laboratory tests and genetic analysis confirms the diagnosis.
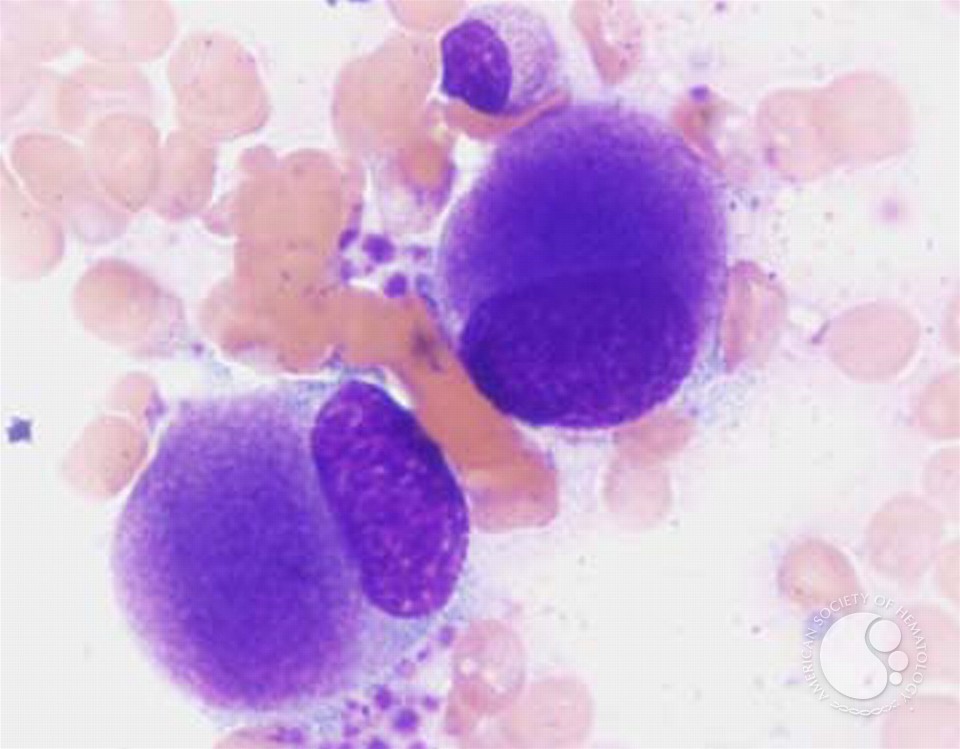
1. Complete Blood Count (CBC) and Peripheral Blood Smear
- Macrocytic anemia with normal or high platelet count.
- Dysplastic red blood cells and megakaryocytes.
2. Bone Marrow Biopsy and Aspirate
- Hypercellular or normocellular marrow with erythroid dysplasia.
- Presence of hypolobulated megakaryocytes.
- Blasts <5%, distinguishing it from high-risk MDS.
3. Cytogenetic and Molecular Testing
- Fluorescence in Situ Hybridization (FISH) or Karyotyping detects 5q deletion.
- Absence of complex karyotypic abnormalities suggests better prognosis.
Risk Stratification in del(5q) MDS
MDS with isolated del(5q) generally falls into the low-risk category based on the Revised International Prognostic Scoring System (IPSS-R):
| Risk Factor | Del(5q) MDS Impact |
|---|---|
| Blast Percentage | Usually <5% (low risk) |
| Cytogenetics | Isolated del(5q) = favorable |
| Cytopenias | Primarily anemia |
| Prognosis | Median survival: >7 years |
Treatment Options for Myelodysplastic Syndrome with 5q Deletion
1. Lenalidomide: Targeted Therapy
- Mechanism: Suppresses abnormal del(5q) clone while stimulating erythropoiesis.
- Efficacy: Achieves transfusion independence in >60% of patients.
- Side Effects: Neutropenia and thrombocytopenia require monitoring.
2. Supportive Care
- Red Blood Cell Transfusions – Manage severe anemia.
- Erythropoiesis-Stimulating Agents (ESAs) – Used in non-responders to lenalidomide.
- Iron Chelation Therapy – Prevents iron overload from frequent transfusions.
3. Hematopoietic Stem Cell Transplantation (HSCT)
- Curative option but reserved for younger, high-risk patients.
- Rarely needed in isolated del(5q) due to favorable prognosis.
Prognosis and Survival in del(5q) MDS
MDS with isolated 5q deletion has one of the best outcomes among MDS subtypes:
- Overall Median Survival: >7 years.
- Risk of AML Transformation: <10% over 5 years.
- Lenalidomide Responders: Improved survival and quality of life.
Myelodysplastic syndrome with 5q deletion is a unique, low-risk MDS subtype with distinct clinical features and a well-defined treatment approach. Targeted therapy with lenalidomide has revolutionized management, significantly improving outcomes. Early diagnosis and appropriate intervention remain key in optimizing patient prognosis.